Precision Kinase-Targeted Therapeutics
Developing disease-modifying
medicines where they're
needed most
TenaRx is advancing a new class of highly selective small-molecule therapies targeting DYRK and CLK kinases — unlocking disease-modifying potential across diabetes, oncology, and neurodegeneration.
Who We Are
Purpose-Built for
Precision Medicine
TenaRx is a San Diego-based biotechnology company developing first-in-class, highly selective small-molecule medicines that target the underlying biology of serious diseases — not just their symptoms.
Our focus is a family of enzymes called DYRK and CLK kinases, which play central roles in immune signaling, cellular regeneration, and RNA processing. While these targets have long been recognized as therapeutically important, prior drug development efforts were limited by a lack of selectivity — early compounds hit too many unintended targets, causing toxicity.
TenaRx was purpose-built to solve that problem. Drawing on more than a decade of dedicated research, we have developed highly selective inhibitors that engage these kinases with precision — unlocking disease-modifying potential while minimizing unwanted side effects.
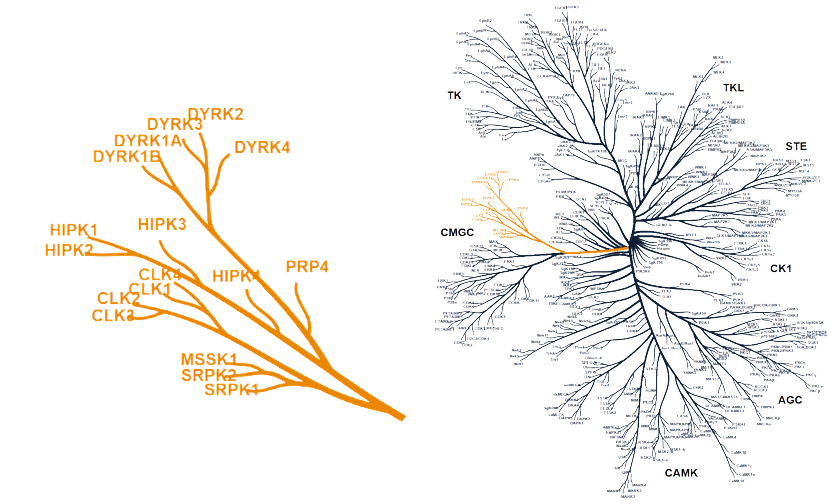
DYRK and CLK kinases highlighted within the human kinome — TenaRx's focus area
Selective Chemistry
Our proprietary medicinal chemistry toolkit has produced drug candidates that precisely target specific members of the DYRK/CLK kinase family while avoiding unintended proteins — enabling a broader therapeutic window.
Disease Modification
We target the root biological drivers of disease — from the loss of insulin-producing cells in diabetes, to the immune evasion mechanisms that let tumors grow unchecked.
Oral Small Molecules
All our therapies are designed as oral drugs — scalable to manufacture, convenient to administer, and suitable for chronic disease settings. Select candidates are also brain-penetrant.
Our Science
A Target Class With Deep Biological Evidence
DYRK and CLK kinases keep appearing in the same places — Alzheimer's literature, diabetes research, cancer biology. Not because of a single hypothesis, but because independent labs across two decades keep finding them at the center of the same broken pathways.
The Biology
Kinases are molecular switches — enzymes that turn proteins on or off by tagging them with a phosphate group. DYRK and CLK are a particular subset that have drawn sustained scientific attention because of what they control:
Before a gene becomes a protein, the cell must "edit" its instructions — a process called RNA splicing. CLK kinases are the master switches of this editing. In breast cancer, CLK2 amplification corrupts splicing to generate oncoproteins that drive tumor growth. Block CLK, and that program breaks down.
Yoshida et al., Cancer Research 2015 →Human pancreatic β cells — the cells that make insulin — divide at less than 0.1% per day in adults. That's why diabetes is so hard to reverse. DYRK1A inhibition changes that: in published human islet studies, combining a DYRK1A inhibitor with a GLP-1 agonist drove β-cell replication rates of 5–6% per day. For a cell type once thought non-renewable, that result is striking.
Ackeifi et al., Science Translational Medicine 2020 →People with Down syndrome carry a third copy of chromosome 21 — which means a third copy of DYRK1A — and develop Alzheimer's pathology at near-100% rates by middle age. DYRK1A directly phosphorylates tau at sites that trigger neurofibrillary tangle formation. In animal models, DYRK1A inhibitors rescued both tau- and amyloid-driven neurodegeneration. This genetic link gives the target unusually strong biological rationale in a disease where most approaches have failed.
Zhu et al., Scientific Reports 2022 →The Challenge — and Our Solution
The human body contains over 500 kinases with similar structures. Early attempts to inhibit DYRK and CLK kinases hit many of them indiscriminately, causing toxicity that limited usefulness in patients.
TenaRx's proprietary medicinal chemistry toolkit has produced multiple distinct drug candidates — each precisely tuned to hit DYRK or CLK targets while leaving unrelated kinases alone. This selectivity makes chronic dosing and multi-indication development possible.
Four programs across diabetes, oncology, and neurodegeneration — built on the same chemistry.
Every program includes measurable biomarkers to confirm target engagement early.
Small-molecule drugs taken orally — no complex biologics or cell therapies.
Built on 10+ years of internal discovery and optimization — not early-stage speculation.
Pipeline
Multiple Programs, One Target Class
Four therapeutic programs across three disease areas — all built on the same core DYRK/CLK chemistry.
DYRK1A Inhibition
Type 1 & Type 2 Diabetes
Stimulates controlled regeneration of insulin-producing β-cells in the pancreas, aiming to restore the body's own insulin production.
Immuno-Oncology Program
Undisclosed
A novel kinase-targeted approach to immuno-oncology. Program details to be disclosed as the program advances.
Cirtuvivint
AML / MDS · Ovarian · SCLC · Sarcoma
Modulates aberrant RNA splicing in cancer cells, reducing production of the abnormal proteins that drive tumor growth and survival.
DYRK1A CNS Program
Alzheimer's Disease & Tauopathies
A brain-penetrant inhibitor designed to reduce pathological tau protein buildup and neuroinflammation in neurodegeneration.
Our Programs
Program Details
DYRK1A Inhibition for β-Cell Regeneration
Type 1 and Type 2 diabetes both converge on the same problem: a deficit of functional insulin-producing β-cells. Our DYRK1A inhibitor program aims to address that deficit directly — by making β-cells grow back.
The Biology
Human pancreatic β-cells barely divide in adults — fewer than 0.1% per day. DYRK1A is a key brake on that process. When it's inhibited, cells re-enter the cell cycle via NFAT nuclear translocation. In published human islet studies, combining a DYRK1A inhibitor with a GLP-1 receptor agonist drove β-cell replication rates of 5–6% per day — a result that surprised even the field.
Our Approach
TenaRx's DYRK1A inhibitor is designed for oral dosing with high selectivity across the kinome — avoiding the off-target toxicity that limited earlier inhibitors. Lead optimization is complete. IND-enabling studies are underway, targeting both T1D (autoimmune β-cell destruction) and T2D (progressive β-cell exhaustion).
Evidence Base
Publications & Presentations
Modulation of RNA Splicing Enhances Response to BCL2 Inhibition
Cancer Cell, 2023
Read publication →Preliminary Evidence of Clinical Activity of Cirtuvivint in Advanced Solid Tumors
ESMO Congress, 2022
View presentation →Multigenomic Characterization of Alternative Splicing
AACR-NCI-EORTC International Conference on Molecular Targets, 2021
View poster →Preclinical Efficacy Landscape of CLK/DYRK Inhibitor Cirtuvivint
AACR-NCI-EORTC International Conference on Molecular Targets, 2021
View poster →SM08502 Antitumor Effects in Castration-Resistant Prostate Cancer Models
AACR Annual Meeting, 2020
View poster →Transcriptome Analysis of CLK Expression and Survival in Prostate Cancer (TCGA)
AACR Annual Meeting, 2020
View poster →CLK Inhibitor Antitumor Activity in Gastrointestinal Cancers
Cancer Letters, 2019
Read publication →Discovery and Preclinical Development of SM15685, a Novel DYRK1A/B Inhibitor for Alzheimer's Disease
Alzheimer's Association International Conference (AAIC), 2024
View poster →Tau Pathology Reduction with SM07883 in Preclinical Models of Neurodegeneration
Aging Cell, 2019
Read publication →Ultra-selective DYRK1A/B Inhibitors for β-Cell Proliferation: In Vitro and In Vivo Studies
American Diabetes Association Scientific Sessions, 2024
View poster →Leadership
Leadership Team
Our leadership has worked together for years within this target class — collectively opening six INDs and conducting more than 15 clinical trials. They are supported by a deep team of drug discovery scientists in chemistry, biology, and preclinical development.

Erich Horsley
Chief Executive Officer
More than a decade of biotech leadership spanning CEO, CFO, and CBO roles. Prior career as a founding member of Intervale Capital, a $1.2 billion private equity firm, and investment banker at Morgan Stanley.

Phil Wilson
Chief Financial Officer
Nearly a decade in biotech business development and finance. Former VP at Oaktree Capital Management and Intervale Capital. Extensive experience closing complex transactions.

Yusuf Yazici, MD
Chief Medical Officer
Rheumatologist and Clinical Associate Professor at NYU with 12 years of clinical development and regulatory experience. Led the first NDA submission for a potentially disease-modifying therapy in osteoarthritis. Author of 250+ publications.

John Hill, PhD
Chief Scientific Officer
Over 25 years in preclinical and early clinical drug development with deep expertise in oncology and diabetes at Amgen, Pharmacyclics, AbbVie, J&J, and Pfizer. Secured $50M+ in non-dilutive funding.

Erik Gutierrez, PhD
Head of Business Development
Over 15 years in biopharma spanning business development, corporate strategy, and discovery research at Esperion, Alexion, Biogen, and Novartis. Executed transactions totaling $1.5B+ in potential value.

Scott Bulcao
General Counsel
More than 15 years advising life sciences and corporate clients on legal matters. Former associate at Simpson Thacher & Bartlett and Schindler Cohen & Hochman in New York. Served a federal judicial clerkship in the Northern District of Texas.
Scientific Advisors
Leading Academics & Clinicians
TenaRx is advised by recognized experts at top academic medical centers across oncology, diabetes, and drug development.
Mark Chao, MD, PhD
Chief Medical Officer · Antares Therapeutics · Co-Founder, Forty Seven, Inc.
20+ years in oncology R&D. Co-founded TenSixteen Bio and Forty Seven, Inc., and served as VP of Oncology Clinical Research at Gilead Sciences. Recognized as a Next-Gen Biotech Leader (Endpoints News, 2021). Deep expertise in immuno-oncology, clinical trial design, and advancing early- to late-stage oncology programs.
Rohit Kulkarni, MD, PhD
Professor of Medicine · Harvard Medical School · Co-Head, Islet & Regenerative Biology, Joslin Diabetes Center
Expert in islet cell growth, signaling, and β-cell regeneration in Type 1 and Type 2 diabetes. Utilizes genetic models, iPS cells, and transplantation studies to advance disease modeling and drug testing. Recipient of the Endocrine Society's Ernst Oppenheimer Award; elected to ASCI and AAP.
Kathleen Moore, MD, MS
Virginia Kerley Cade Endowed Chair in Cancer Development · University of Oklahoma · Stephenson Cancer Center
Fellowship-trained gynecologic oncologist and Director of the Oklahoma TSET Phase I Program. Expert in new drug development, Phase I clinical trials, and translational oncology. Program Director of the Gynecologic Oncology Fellowship Program.